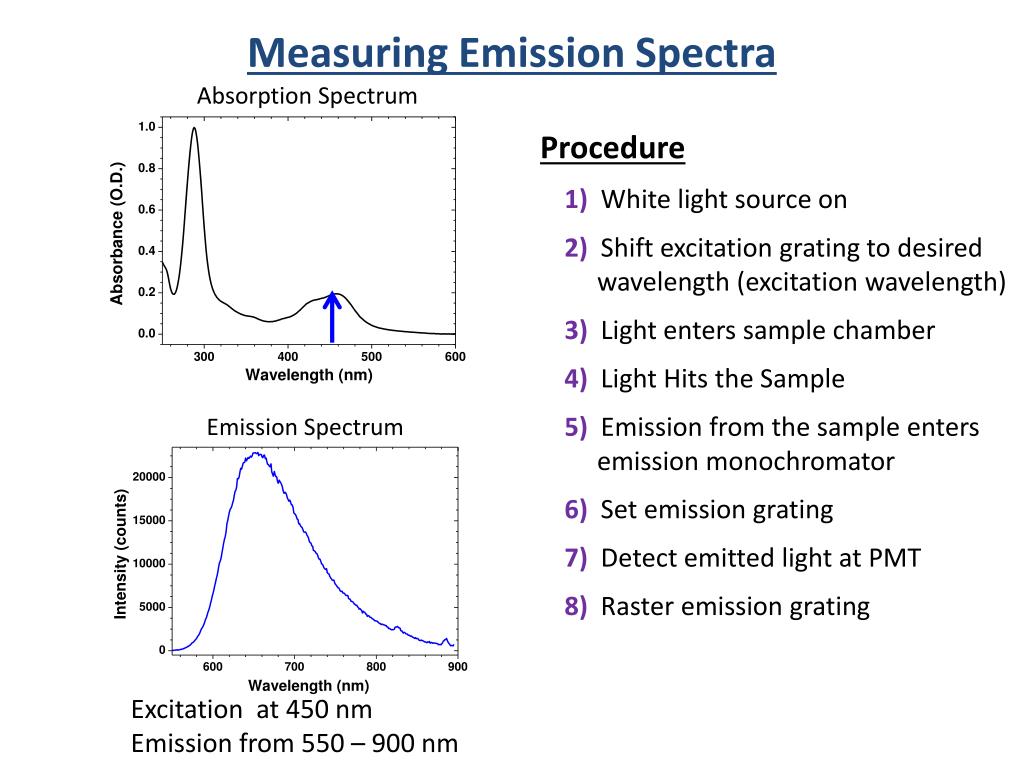
  Therefore, when this light passes through a gas, the gas atoms may absorb certain wavelengths to produce a line absorption spectrum.īlack hydrogen absorption lines are at the same wavelength as the bright hydrogen emission lines. Stokes shift is the difference (in energy, wavenumber or frequency units) between positions of the band maxima of the absorption and emission spectra (fluorescence and Raman being two examples) of the same electronic transition. This is called a line absorption spectrum. Emission spectra help identify elements or compounds in a sample, whereas absorption spectra provide information about a substance’s chemical structure and concentration. DIFFERENCE BETWEEN EMISSION AND ABSORPTION SPECTRA SERIESSo if a continuous spectrum travels through a cooler gas, it will ‘pull out’ or absorb certain wavelengths of light to produce a series of dark lines on a continuous spectrum of light. These absorption and emission spectra show relative intensity of fluorescence, with the relative intensity classically plotted on the vertical axis versus wavelength on the horizontal axis. When a gas is cool, it absorbs the same wavelengths of light as it would emit when it is hot. Knox, On the relation between absorption and emission spectra of molecules in solution. Absorbance log(P0/P) Beer’s Law: A bc b pathlength (in cm) c concentration (in M) molar absorptivity (units of M-1cm-1) Absorption is the process that consumes a photon and puts the atom or molecule in an excited state. A gas cloud on its own, without a light source behind it, produces a line emission spectrum. Fine differences between pump and probe paths in the 2D interferometer make their pulse spectra slightly different. Hot gases don’t produce a continuous emission spectrum.Ī hot gas only emits certain wavelengths of light to produce bright lines on a dark background. When a gas is very hot, it doesn’t emit all wavelengths of light. The amount of energy it loses will be equal to the difference in. A light source, such as a star or a filament bulb, gives a continuous emission spectrum. If an electron is in an excited state it can return to a lower energy level. This is called a continuous emission spectrum. Emission and Absorption Spectra Differences in Physics Import Concepts Must to Know Energy of Atom, An emission spectrum is given when an excited atom obtains. This light, when seen through a prism or diffraction grating, shows all wavelengths of visible light. Studying the line spectra produced by hot gases and absorbed by cooler gases allows us to identify the elements in stars. Absorption spectra (also known as UV-Vis spectra, absorbance spectra and electronic spectra) show the change in absorbance of a sample as a function of the. 
0 Comments
Leave a Reply. |
AuthorWrite something about yourself. No need to be fancy, just an overview. ArchivesCategories |
 RSS Feed
RSS Feed
